Acta Agriculturae Zhejiangensis ›› 2026, Vol. 38 ›› Issue (1): 54-66.DOI: 10.3969/j.issn.1004-1524.20250366
• Horticultural Science • Previous Articles Next Articles
The influence of Rhizophagus irregularis on the growth and gene expression of tomato under cadmium stress
LIU Junli1( ), JIANG Jianfeng2, DONG Xiangwei2, YANG Haijun2, BAO Xiaoqi1,3, FU Chenxi1, GUO Bin1, TONG Wenbin2,*(
), JIANG Jianfeng2, DONG Xiangwei2, YANG Haijun2, BAO Xiaoqi1,3, FU Chenxi1, GUO Bin1, TONG Wenbin2,*( )
)
- 1. Institute of Environment, Resource, Soil and Fertilizer, State Key Laboratory for Quality and Safety of Agro-Products, Zhejiang Academy of Agricultural Sciences, Hangzhou 310021, China
2. Qujiang District Agricultural Technology Promotion Center, Quzhou 324022, Zhejiang, China
3. College of Environment, Zhejiang University of Technology, Hangzhou 310014, China
-
Received:2025-03-14Online:2026-01-25Published:2026-02-11
CLC Number:
Cite this article
LIU Junli, JIANG Jianfeng, DONG Xiangwei, YANG Haijun, BAO Xiaoqi, FU Chenxi, GUO Bin, TONG Wenbin. The influence of Rhizophagus irregularis on the growth and gene expression of tomato under cadmium stress[J]. Acta Agriculturae Zhejiangensis, 2026, 38(1): 54-66.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://www.zjnyxb.cn/EN/10.3969/j.issn.1004-1524.20250366
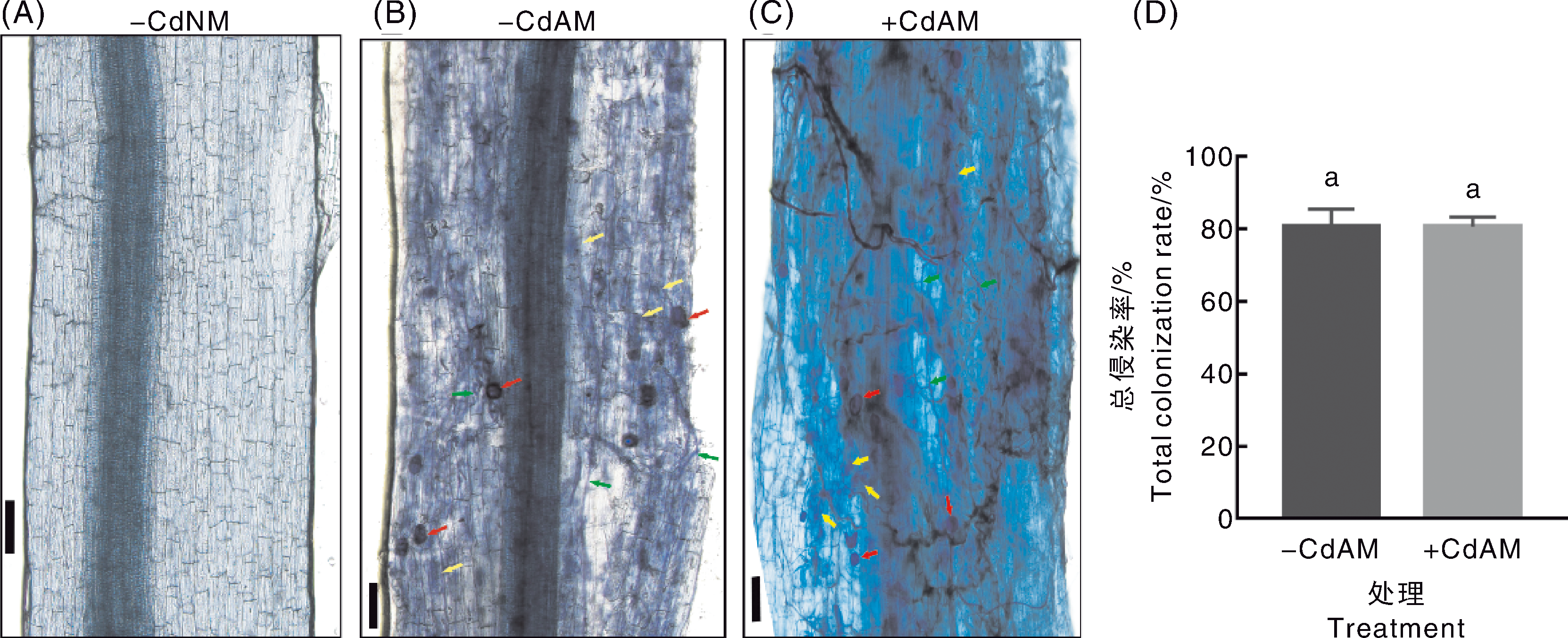
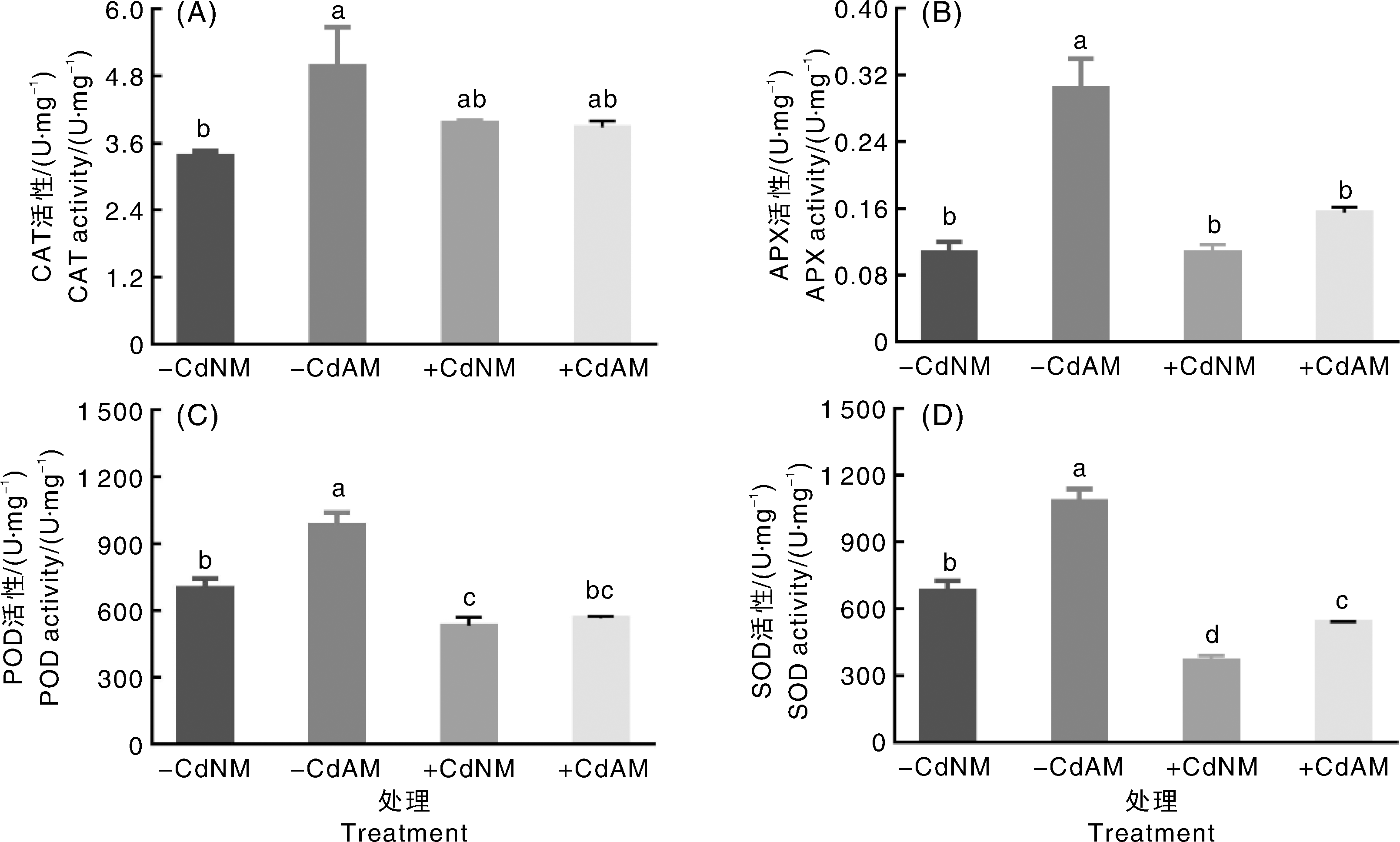
Fig.1 Symbiotic relationship between arbuscular mycorrhizal fungi and tomato under cadmium stress A-C, Microscopic observation of structures such as hyphae, vesicles and arbuscular branches in tomato roots; red arrows indicate vesicles, yellow arrows indicate arbuscular branches, green arrows indicate extraradical hyphae, scale bar=100 μm. D, Total colonization rate of tomato roots; data were mean±standard error (n=3), data marked without the same lowercase letter indicated significant differences (p<0.05, Duncan’s test).-CdNM, no Cd added, no mycorrhizal fungi inoculated;-CdAM, no Cd added, mycorrhizal fungi inoculated;+CdAM, Cd added, mycorrhizal fungi inoculated. The same as below.
| 处理 Teatment | 根部生物量/g Root biomass/g | 地上部生物量/g Shoot biomass/g | 根冠比 Root shoot ratio |
|---|---|---|---|
| -CdNM | 0.014±0.001 b | 0.221±0.011 b | 0.061±0.003 a |
| -CdAM | 0.020±0.001 a | 0.304±0.028 a | 0.067±0.004 a |
| +CdNM | 0.014±0.002 b | 0.164±0.016 c | 0.089±0.022 a |
| +CdAM | 0.012±0.001 b | 0.208±0.002 b | 0.058±0.004 a |
Table 1 Biomass (dry weight) per plant of tomato under different treatments
| 处理 Teatment | 根部生物量/g Root biomass/g | 地上部生物量/g Shoot biomass/g | 根冠比 Root shoot ratio |
|---|---|---|---|
| -CdNM | 0.014±0.001 b | 0.221±0.011 b | 0.061±0.003 a |
| -CdAM | 0.020±0.001 a | 0.304±0.028 a | 0.067±0.004 a |
| +CdNM | 0.014±0.002 b | 0.164±0.016 c | 0.089±0.022 a |
| +CdAM | 0.012±0.001 b | 0.208±0.002 b | 0.058±0.004 a |
| 处理 Teatment | 根部Cd含量/ (mg·g-1) Cd content in root/(mg·g-1) | 地上部Cd含量/ (mg·g-1) Cd content in shoot/(mg·g-1) | 镉转运系数 Transfer coefficient of cadmium |
|---|---|---|---|
| -CdNM | 0.004±0 c | 0.001±0 c | — |
| -CdAM | 0.003±0.001 c | 0.001±0 c | — |
| +CdNM | 3.473±0.530 a | 0.118±0.013 a | 0.035±0.003 a |
| +CdAM | 2.073±0.236 b | 0.073±0.007 b | 0.036±0.001 a |
Table 2 Cd content and transfer coefficient in tomato plants under different treatments
| 处理 Teatment | 根部Cd含量/ (mg·g-1) Cd content in root/(mg·g-1) | 地上部Cd含量/ (mg·g-1) Cd content in shoot/(mg·g-1) | 镉转运系数 Transfer coefficient of cadmium |
|---|---|---|---|
| -CdNM | 0.004±0 c | 0.001±0 c | — |
| -CdAM | 0.003±0.001 c | 0.001±0 c | — |
| +CdNM | 3.473±0.530 a | 0.118±0.013 a | 0.035±0.003 a |
| +CdAM | 2.073±0.236 b | 0.073±0.007 b | 0.036±0.001 a |
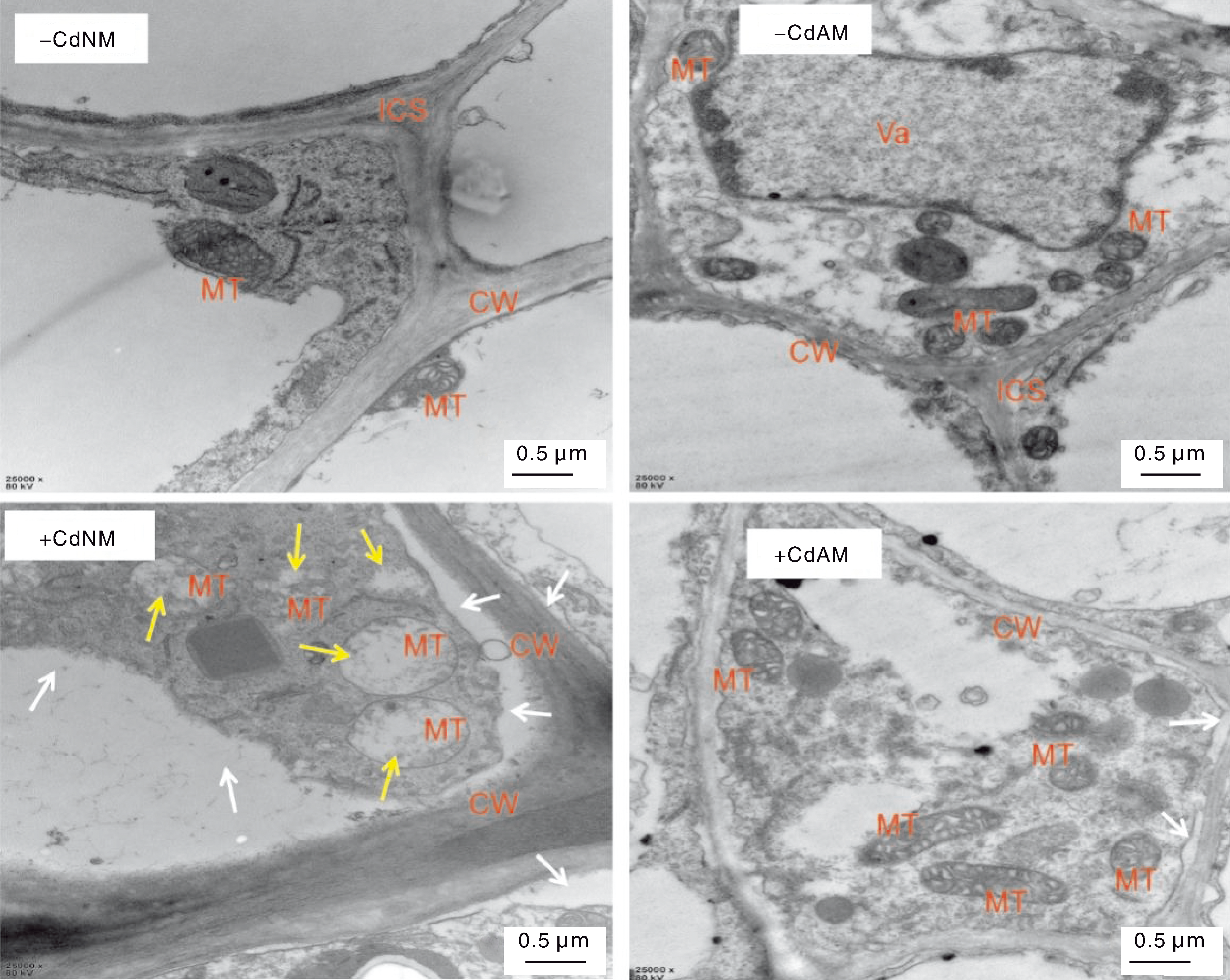
Fig.3 Ultrastructure of tomato root tissue cells under different treatments MT, Mitochondria; CW, Cell wall; ICS, Intercellular space; Va, Vacuole. White arrows indicate plasmolysis, yellow arrows indicate mitochondrial vacuolization.
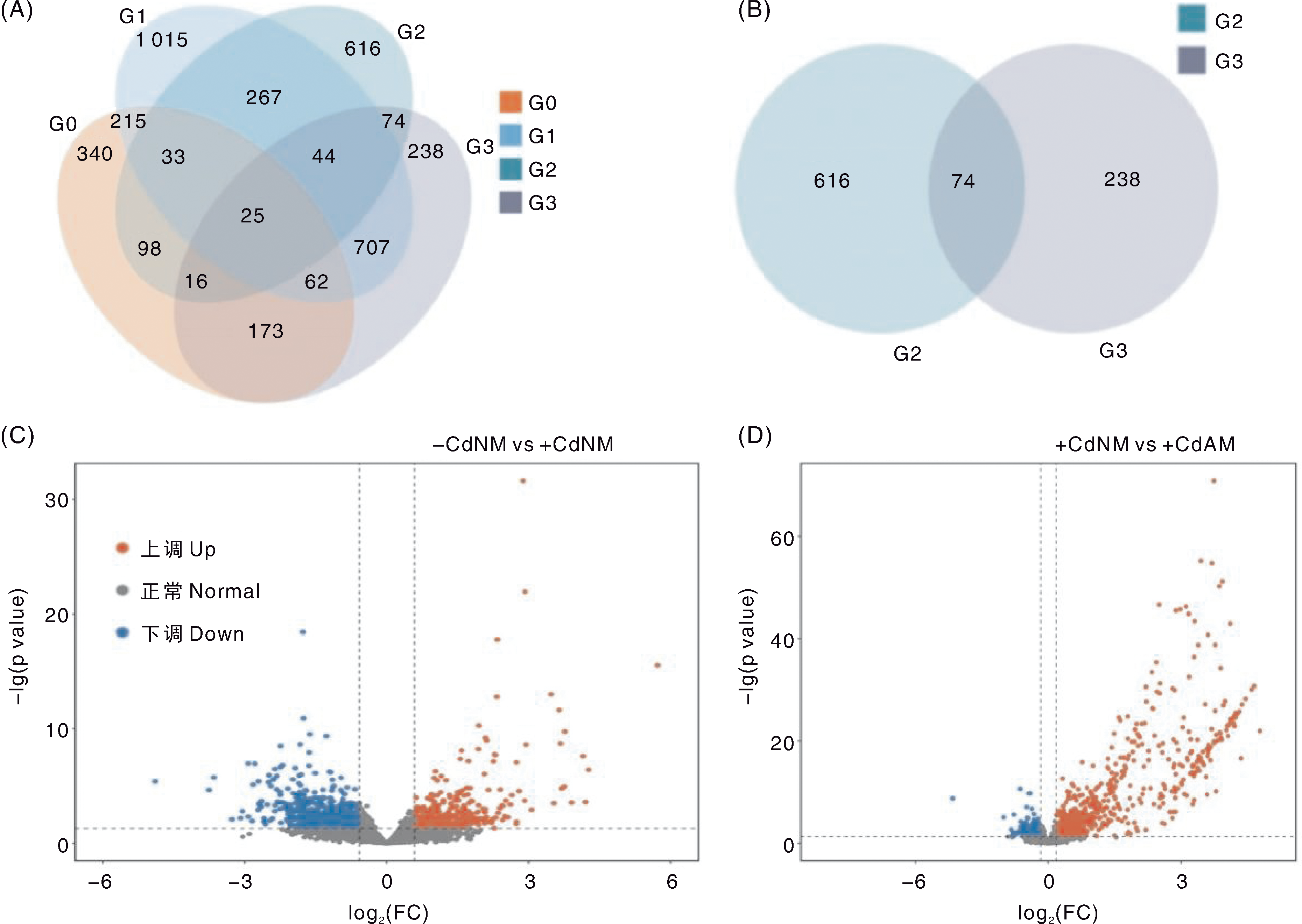
Fig.4 Differentially expressed genes in tomato roots under different treatments A, Venn diagram of differentially expressed genes in tomato roots under different treatments; G0, G1, G2, and G3 represent differentially expressed genes between the comparison groups+CdAM vs-CdAM,-CdAM vs-CdNM,+CdNM vs-CdNM, and+CdAM vs+CdNM, respectively. B, Venn diagram of differentially expressed genes in the two comparison groups G2 and G3. C and D are gene expression volcano plots for the comparison groups-CdNM vs+CdNM and+CdNM vs+CdAM, respectively. FC, Fold change.
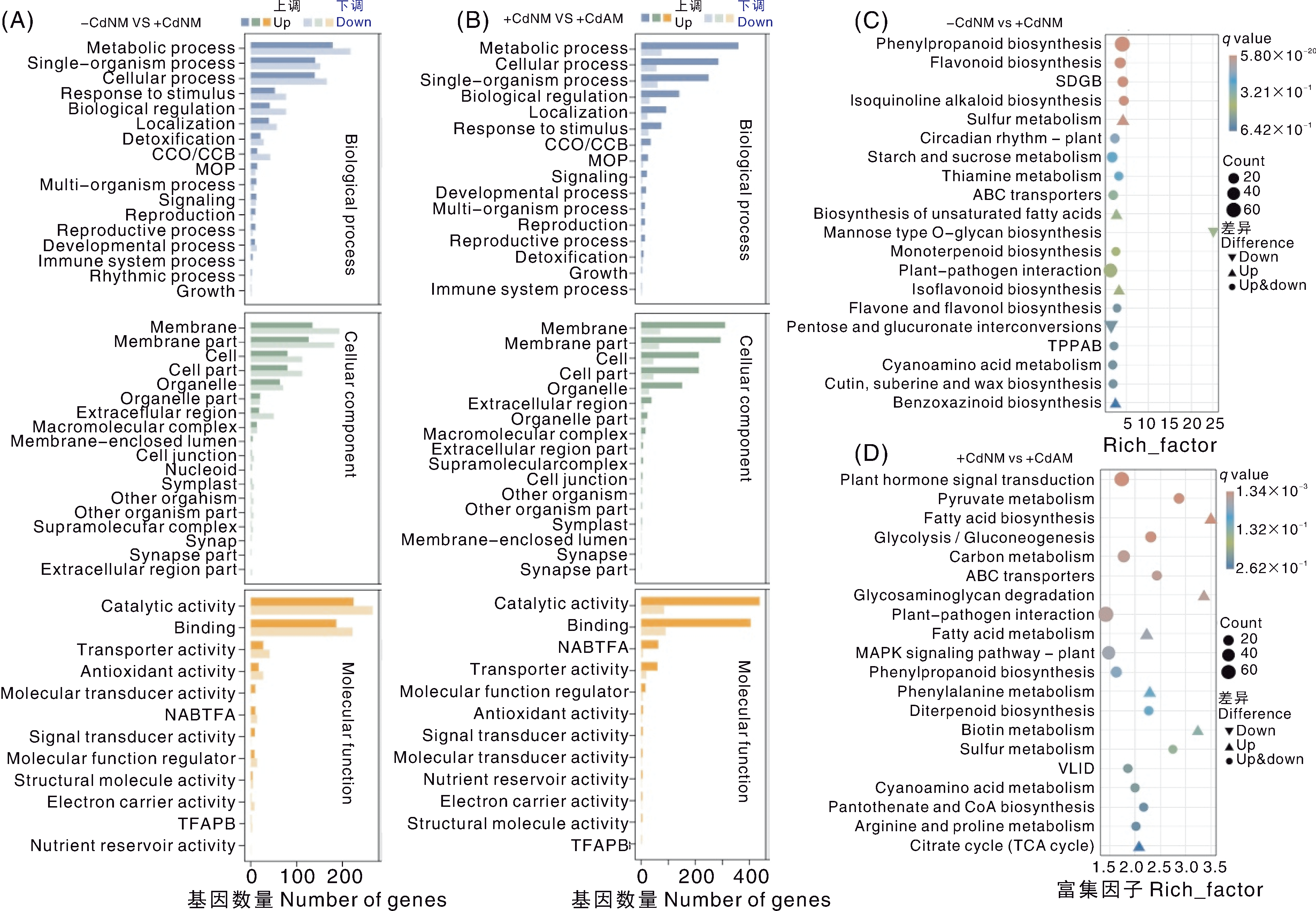
Fig.5 GO and KEGG enrichment analysis of differentially expressed genes A and B, Gene Ontology (GO) enrichment analysis of differentially expressed genes (DEGs) from the comparison groups -CdNM vs +CdNM and +CdNM vs +CdAM, respectively. CCO/CCB, Cellular component organization or biogenesis; MOP, Multicellular organismal process; NABTFA, Nucleic acid binding transcription factor activity; TFAPB, Transcription factor activity, protein binding. C and D, KEGG pathway enrichment analysis of DEGs from the same comparison groups. The figure shows the top 20 pathways with the smallest q-values. TPPAB, Tropane, piperidine and pyridine alkaloid biosynthesis; SDGB, Stilbenoid, diarylheptanoid and gingerol biosynthesis; VLID, Valine, leucine and isoleucine degradation.
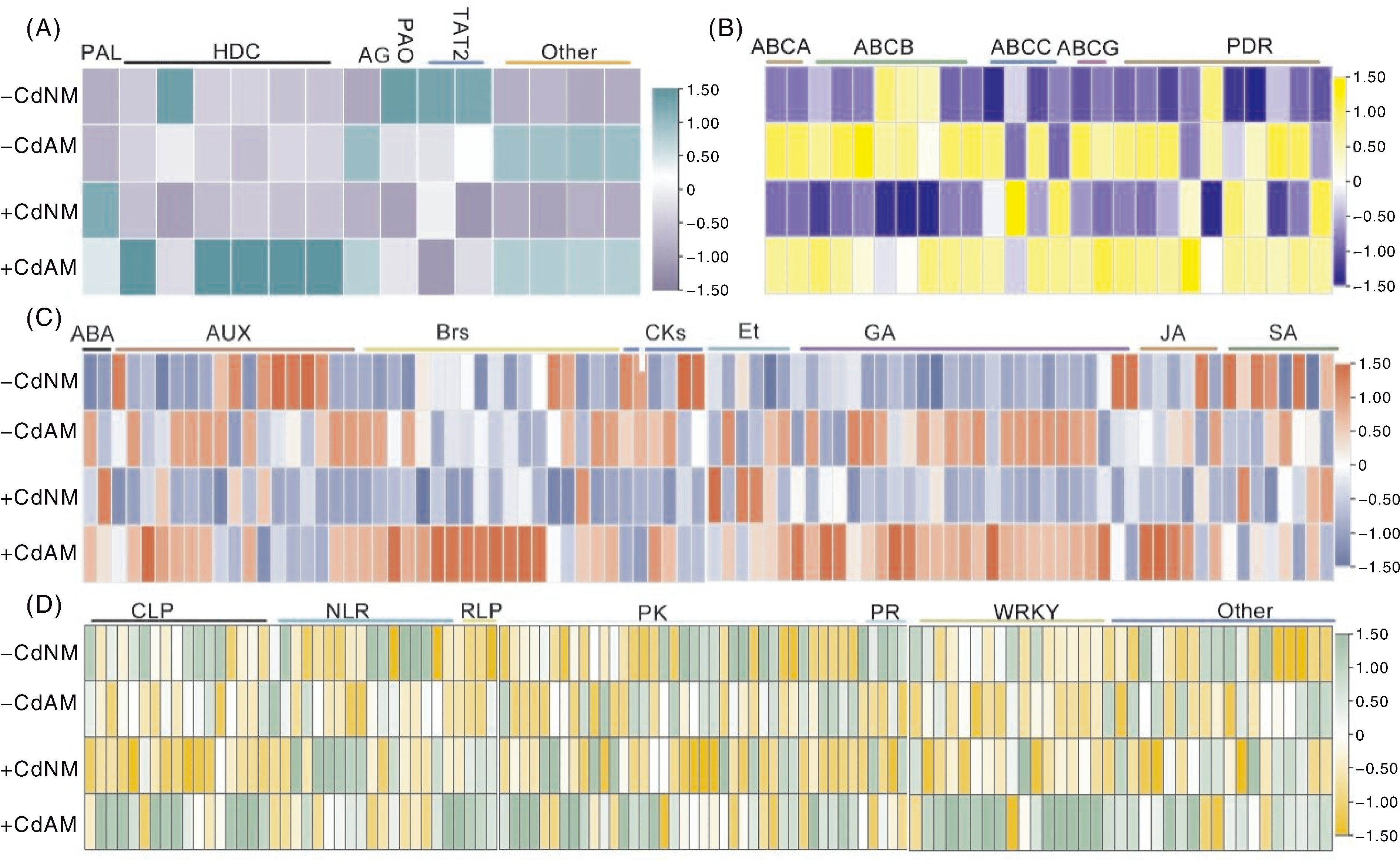
Fig.6 Heatmap of differentially expressed genes related to phenylalanine metabolism (A), ABC transporter family gene (B), plant hormone signal transduction (C) and plant-pathogen interaction (D) A, Heatmap of differentially expressed genes (DEGs) related to phenylalanine metabolism pathway. PAL, Phenylalanine ammonia-lyase gene; HDC, Histidine decarboxylase gene; AG, Amidase gene; PAO, Primary amine oxidase gene; TAT2, Aminotransferase gene; Other, Other genes. B, Heatmap of DEGs associated with ABC transporter family genes. ABCA/B/C/G represent subfamilies A, B, C and G of ABC transporter family genes, respectively; PDR, Pleiotropic drug resistance gene (belonging to ABCG subfamily). C, Heatmap of DEGs involved in plant hormone signal transduction. ABA, Abscisic acid gene; AUX, Auxin gene; Brs, Brassinosteroids gene; CK, Cytokinins gene; Et, Ethylene gene; GA, Gibberellins gene; JA, Jasmonic acid gene; SA, Salicylic acid gene. D, Heatmap of DEGs related to plant-pathogen interaction. CLP, Calmodulin-like protein gene; NLR, NLR family gene; RLP, Receptor-like protein gene; PK, Protein kinase gene; PR, Pathogenesis-related protein gene; WRKY, WRKY transcription factor gene; Other, Other genes. Data are presented as log2|FPKM mean+1| (n=3), the scale bar is the numerical range corresponding to the color in the heatmap.
| [1] | 翟夜雨, 黄五星, 袁岐山, 等. 植物镉毒害与硒对镉胁迫的缓解作用研究进展[J]. 河南农业大学学报, 2023, 57(3): 372-382. |
| ZHAI Y Y, HUANG W X, YUAN Q S, et al. Research progress on plant cadmium toxicity and the alleviation effect of selenium on cadmium stress[J]. Journal of Henan Agricultural University, 2023, 57(3): 372-382. | |
| [2] | 陈瑾芬, 胡淑宝, 秦艺鸣, 等. 植物对镉胁迫响应的分子机制研究进展[J]. 安徽农业科学, 2025, 53(3): 1-9. |
| CHEN J F, HU S B, QIN Y M, et al. Research progress on the molecular mechanism of plant response to cadmium stress[J]. Journal of Anhui Agricultural Sciences, 2025, 53(3): 1-9. | |
| [3] | 彭佳师, 王娅婷, 王梦琦, 等. 植物重金属镉积累调控机制及其应用研究进展[J]. 植物生理学报, 2024, 60(2): 185-210. |
| PENG J S, WANG Y T, WANG M Q, et al. Research and regulation of cadmium uptake, transport and accumulation in plants[J]. Plant Physiology Journal, 2024, 60(2): 185-210. | |
| [4] | 李娜, 王剑峰, 龚记熠, 等. 植物响应镉胁迫的生理、分子和根际微生态机制研究进展[J]. 江苏农业科学, 2024, 52(24): 1-11. |
| LI N, WANG J F, GONG J Y, et al. Research progress on physiological, molecular and rhizosphere microecological mechanisms of plant response to cadmium stress[J]. Jiangsu Agricultural Sciences, 2024, 52(24): 1-11. | |
| [5] | 段世龙, 严文辉, 冯固, 等. 植物根系/菌根途径获取养分的碳磷互惠机制[J]. 植物营养与肥料学报, 2023, 29(6): 1160-1167. |
| DUAN S L, YAN W H, FENG G, et al. Carbon-phosphorus reciprocal mechanism for plants to acquire nutrients through the root/mycorrhizal pathway[J]. Journal of Plant Nutrition and Fertilizers, 2023, 29(6): 1160-1167. | |
| [6] | LI X, ZHENG D, ZHANG N L, et al. Arbuscular mycorrhizal fungi-mediated resistance to salt spray in Cinnamomum camphora seedlings enhanced by leaf functional traits[J]. Soil Ecology Letters, 2023, 6(3): 230211. |
| [7] | 张嘉慧, 邢佳佳, 彭丽媛, 等. 丛枝菌根真菌提高感染青枯菌番茄根际土壤细菌群落多样性和稳定性及有益菌属相对丰度[J]. 植物营养与肥料学报, 2023, 29(1): 120-131. |
| ZHANG J H, XING J J, PENG L Y, et al. Arbuscular mycorrhizal fungi improves diversity and stability of bacterial community and abundance of beneficial bacteria genus in the rhizosphere of tomato infected with Ralstonia solanacearum[J]. Journal of Plant Nutrition and Fertilizers, 2023, 29(1): 120-131. | |
| [8] | JIN W H, TU J Y, WU Q F, et al. Moso bamboo expansion decreased soil heterotrophic respiration but increased arbuscular mycorrhizal mycelial respiration in a subtropical broadleaved forest[J]. Forest Ecosystems, 2023, 10: 100116. |
| [9] | WANG Y H, SHAO C L, QIU Y J, et al. Arbuscular mycorrhizal fungi protect a subtropical tree species exposed to simulated acid rain by accelerating photosynthetic ability, antioxidant enzymes and osmolyte accumulation[J]. Journal of Plant Ecology, 2022, 15(5): 1036-1048. |
| [10] | SHI W G, ZHANG Y H, CHEN S L, et al. Physiological and molecular mechanisms of heavy metal accumulation in nonmycorrhizal versus mycorrhizal plants[J]. Plant, Cell & Environment, 2019, 42(4): 1087-1103. |
| [11] | CHEN B D, NAYUKI K, KUGA Y, et al. Uptake and intraradical immobilization of cadmium by arbuscular mycorrhizal fungi as revealed by a stable isotope tracer and synchrotron radiation μX-ray fluorescence analysis[J]. Microbes and Environments, 2018, 33(3): 257-263. |
| [12] | ZHANG Q M, GONG M G, XU S S, et al. Arbuscular mycorrhizal fungi alleviate arsenic toxicity in Sophora viciifolia Hance. by improving the growth, photosynthesis, reactive oxygen species and gene expression of phytochelatin synthase[EB/OL]. ( 2021-01-04)[2025-01-20]. https://doi.org/10.21203/rs.3.rs-137602/v1. |
| [13] | ZHANG X F, HU Z H, YAN T X, et al. Arbuscular mycorrhizal fungi alleviate Cd phytotoxicity by altering Cd subcellular distribution and chemical forms in Zea mays[J]. Ecotoxicology and Environmental Safety, 2019, 171: 352-360. |
| [14] | ZHUANG X L, LIU S Y, XU S Z, et al. Arbuscular mycorrhizal fungi alleviate cadmium phytotoxicity by regulating cadmium mobility, physiological responses, and gene expression patterns in Malus hupehensis Rehd[J]. International Journal of Molecular Sciences, 2025, 26(4): 1418. |
| [15] | ZHU Q Y, XU P X, LEI L L, et al. Transcriptome analysis reveals decreased accumulation and toxicity of Cd in upland rice inoculated with arbuscular mycorrhizal fungi[J]. Applied Soil Ecology, 2022, 177: 104501. |
| [16] | AHMED D A E, SLIMA D F, AL-YASI H M, et al. Risk assessment of trace metals in Solanum lycopersicum L. (tomato) grown under wastewater irrigation conditions[J]. Environmental Science and Pollution Research, 2023, 30(14): 42255-42266. |
| [17] | SU L H, XIE Y D, HE Z Q, et al. Network response of two cherry tomato (Lycopersicon esculentum) cultivars to cadmium stress as revealed by transcriptome analysis[J]. Ecotoxicology and Environmental Safety, 2021, 222: 112473. |
| [18] | PAN J, CAO S, XU G F, et al. Comprehensive analysis reveals the underlying mechanism of arbuscular mycorrhizal fungi in kenaf cadmium stress alleviation[J]. Chemosphere, 2023, 314: 137566. |
| [19] | WANG Y P, HUANG J, GAO Y Z. Arbuscular mycorrhizal colonization alters subcellular distribution and chemical forms of cadmium in Medicago sativa L. and resists cadmium toxicity[J]. PLoS One, 2012, 7(11): e48669. |
| [20] | DAS D, PARIES M, HOBECKER K, et al. PHOSPHATE STARVATION RESPONSE transcription factors enable arbuscular mycorrhiza symbiosis[J]. Nature Communications, 2022, 13: 477. |
| [21] | BALZERGUE C, CHABAUD M, BARKER D G, et al. High phosphate reduces host ability to develop arbuscular mycorrhizal symbiosis without affecting root calcium spiking responses to the fungus[J]. Frontiers in Plant Science, 2013, 4: 426. |
| [22] | LIU J L, CHEN J D, XIE K, et al. A mycorrhiza-specific H+-ATPase is essential for arbuscule development and symbiotic phosphate and nitrogen uptake[J]. Plant, Cell & Environment, 2020, 43(4): 1069-1083. |
| [23] | LIU J L, BAO X Q, QIU G Y, et al. Genome-wide identification and expression analysis of SlNRAMP genes in tomato under nutrient deficiency and cadmium stress during arbuscular mycorrhizal symbiosis[J]. International Journal of Molecular Sciences, 2024, 25(15): 8269. |
| [24] | BAO X Q, LIU J L, QIU G Y, et al. The effect of Rhizophagus intraradices on cadmium uptake and OsNRAMP5 gene expression in rice[J]. International Journal of Molecular Sciences, 2025, 26(4): 1464. |
| [25] | 刘月芹, 高小朋, 贺晓龙. 提高温度对灵芝抗氧化活性的影响[J]. 延安大学学报(自然科学版), 2020, 39(4): 27-31. |
| LIU Y Q, GAO X P, HE X L. Effects of increasing temperature on antioxidant activity of Ganoderma lucidum[J]. Journal of Yanan University(Natural Science Edition), 2020, 39(4): 27-31. | |
| [26] | LIU D, ZHENG K Y, WANG Y, et al. Harnessing an arbuscular mycorrhizal fungus to improve the adaptability of a facultative metallophytic poplar (Populus yunnanensis) to cadmium stress: physiological and molecular responses[J]. Journal of Hazardous Materials, 2022, 424: 127430. |
| [27] | 宋西娇, 陈浙, 何步远, 等. 西瓜花叶病毒和小西葫芦黄花叶病毒复合侵染南瓜的透射电镜诊断[J]. 电子显微学报, 2015, 34(2): 126-131. |
| SONG X J, CHEN Z, HE B Y, et al. Electron microscopic diagnosis of multiple pathogen of watermelon mosaic virus and Zucchini yellow mosaic virus in infected Cucurbita moschata[J]. Journal of Chinese Electron Microscopy Society, 2015, 34(2): 126-131. | |
| [28] | 杨慧, 金良韵, 姬曼, 等. 不同树脂对特殊生物样品包埋效果的比较[J]. 分析仪器, 2019(5): 46-51. |
| YANG H, JIN L Y, JI M, et al. Comparison of embedding effects of two resins on special biological specimens[J]. Analytical Instrumentation, 2019(5): 46-51. | |
| [29] | CHEN C J, WU Y, LI J W, et al. TBtools-II: a “one for all, all for one” bioinformatics platform for biological big-data mining[J]. Molecular Plant, 2023, 16(11): 1733-1742. |
| [30] | LUO N, LI X, CHEN A Y, et al. Does arbuscular mycorrhizal fungus affect cadmium uptake and chemical forms in rice at different growth stages[J]. Science of The Total Environment, 2017, 599: 1564-1572. |
| [31] | JIANG Q Y, ZHUO F, LONG S H, et al. Can arbuscular mycorrhizal fungi reduce Cd uptake and alleviate Cd toxicity of Lonicera japonica grown in Cd-added soils[J]. Scientific Reports, 2016, 6: 21805. |
| [32] | LI H, WANG H X, ZHAO J N, et al. Physio-biochemical and transcriptomic features of arbuscular mycorrhizal fungi relieving cadmium stress in wheat[J]. Antioxidants, 2022, 11(12): 2390. |
| [33] | GU L J, ZHAO M L, GE M, et al. Transcriptome analysis reveals comprehensive responses to cadmium stress in maize inoculated with arbuscular mycorrhizal fungi[J]. Ecotoxicology and Environmental Safety, 2019, 186: 109744. |
| [34] | ZHOU Y, FU J Y, YE Y Q, et al. Physiological and molecular response mechanisms of tomato seedlings to cadmium (Cd) and lead (Pb) stress[J]. PeerJ, 2024, 12: e18533. |
| [35] | 刘伟, 樊文华, 刘奋武, 等. 施磷对镉胁迫下黄瓜苗期光合作用及抗氧化酶系统的影响[J]. 土壤通报, 2022, 53(3): 596-604. |
| LIU W, FAN W H, LIU F W, et al. Effects of phosphorus on photosynthesis and antioxidant enzyme system of cucumber seedlings under cadmium stress[J]. Chinese Journal of Soil Science, 2022, 53(3): 596-604. | |
| [36] | TAN X H, WANG D P, ZHANG X W, et al. A pair of LysM receptors mediates symbiosis and immunity discrimination in Marchantia[J]. Cell, 2025, 188(5): 1330-1348. |
| [37] | FU S, LU Y S, ZHANG X, et al. The ABC transporter ABCG36 is required for cadmium tolerance in rice[J]. Journal of Experimental Botany, 2019, 70(20): 5909-5918. |
| [38] | TAKAHASHI R, ISHIMARU Y, SHIMO H, et al. The OsHMA2 transporter is involved in root-to-shoot translocation of Zn and Cd in rice[J]. Plant, Cell & Environment, 2012, 35(11): 1948-1957. |
| [39] | CAO L, LIU L Y, ZHANG C, et al. The MYC2 and MYB43 transcription factors cooperate to repress HMA2 and HMA4 expression, altering cadmium tolerance in Arabidopsis thaliana[J]. Journal of Hazardous Materials, 2024, 479: 135703. |
| [40] | KAUSHIK S, RANJAN A, SINGH A K, et al. Methyl jasmonate reduces cadmium toxicity by enhancing phenol and flavonoid metabolism and activating the antioxidant defense system in pigeon pea (Cajanus cajan)[J]. Chemosphere, 2024, 346: 140681. |
| [41] | LI Y, ZHANG S N, BAO Q L, et al. Jasmonic acid alleviates cadmium toxicity through regulating the antioxidant response and enhancing the chelation of cadmium in rice (Oryza sativa L.)[J]. Environmental Pollution, 2022, 304: 119178. |
| [42] | WANG H R, ZHAO X Y, ZHANG J M, et al. Arbuscular mycorrhizal fungus regulates cadmium accumulation, migration, transport, and tolerance in Medicago sativa[J]. Journal of Hazardous Materials, 2022, 435: 129077. |
| [43] | LIU J L, QIU G Y, LIU C, et al. Salicylic acid, a multifaceted hormone, combats abiotic stresses in plants[J]. Life, 2022, 12(6): 886. |
| [44] | SAPARA K K, KHEDIA J, AGARWAL P, et al. SbMYB15 transcription factor mitigates cadmium and nickel stress in transgenic tobacco by limiting uptake and modulating antioxidative defence system[J]. Functional Plant Biology, 2019, 46(8): 702-714. |
| [1] | XU Yan, LI Sujuan, CHEN Guang, XU Shengchun, WANG Jian. Identification of common genes for salt and cadmium tolerance in soybean [J]. Acta Agriculturae Zhejiangensis, 2026, 38(1): 1-16. |
| [2] | ZHU Changsong, NA Qiting, ZHANG Mengzhuo, CAO Hui, LIU Shiying, ZHANG Zhengke, MENG Lanhuan. Effect of SlCHRC gene on tomato floral thermotolerance under high-temperature environment [J]. Acta Agriculturae Zhejiangensis, 2026, 38(1): 67-75. |
| [3] | PEI Huimin, WU Mingming, ZHAI Rongrong, YE Jing, JIN Yue, ZHU Yi, HOU Jianjun, ZHU Guofu, YE Shenghai. Research progress on gene function and breeding of low-cadmium rice cultivars [J]. Acta Agriculturae Zhejiangensis, 2025, 37(9): 2012-2020. |
| [4] | SHI Yangyang, LYU Lixia, TUO Dengfeng. Effects of AMF and PGPR on growth and nutrient absorption of Matthiola incana under low temperature and weak light stress [J]. Acta Agriculturae Zhejiangensis, 2025, 37(8): 1694-1705. |
| [5] | ZHANG Ruonan, MEN Xiaoming, QIN Kaipeng, WANG Binbin, WU Jie, DING Xiangbin, XU Ziwei, QI Keke. Comparative study on growth performance, carcass quality, meat performance and profitability of different crossbreed combinations of Lvjiahei pigs [J]. Acta Agriculturae Zhejiangensis, 2025, 37(6): 1203-1211. |
| [6] | XIANG Ying, CONG Jianmin, PAN Danhong, TAO Yonggang. Comprehensive evaluation of the growth process of different tomato varieties under spring organic greenhouse planting [J]. Acta Agriculturae Zhejiangensis, 2025, 37(6): 1252-1261. |
| [7] | LIU Pengfei, ZHANG Shuhan, HONG Kai, SHAO Yue, LOU Binggan. Isolation and identification of the pathogen causing tomato canker in Zhejiang Province of China [J]. Acta Agriculturae Zhejiangensis, 2025, 37(6): 1293-1300. |
| [8] | ZOU Junyan, WANG Yunzhu, ZHAO Wanqiu, YIN Zhihao, DU Jianke, SUN Chongbo. Research progress of protocorm and protocorm-like body in Orchidaceae plants [J]. Acta Agriculturae Zhejiangensis, 2025, 37(6): 1372-1389. |
| [9] | SU Yang, SHANG Xiaolan, QIAN Zhongming, WU Lingen, HUANG Jiaqi, ZHUANG Haifeng, ZHAO Yufei, DANG Hongyang, XU Lijun. Effects of synergistic enhancement of straw returning to the field with decomposition agent and biochar on soil quality and rice growth [J]. Acta Agriculturae Zhejiangensis, 2025, 37(5): 1139-1148. |
| [10] | HU Xinrou, WANG Mei, ZHANG Yafen, CAI Weiming, JIN Qunli. Effect of abiotic stress on growth development and response mechanism of Ganoderma [J]. Acta Agriculturae Zhejiangensis, 2025, 37(5): 1182-1190. |
| [11] | DI Yancui, JI Zelin, WANG Yuanyuan, LOU Shihao, ZHANG Tao, GUO Zhixin, SHEN Shunshan, PIAO Fengzhi, DU Nanshan, DONG Xiaoxing, DONG Han. Identification, subcellular localization and expression analysis of tomato SlMYB52 gene [J]. Acta Agriculturae Zhejiangensis, 2025, 37(4): 808-819. |
| [12] | REN Yuanlong, MA Rong, WANG Xiaozhuo, ZHANG Xueyan. Mitigative effect of foliar spraying melatonin on drought stress of cabbage seedlings [J]. Acta Agriculturae Zhejiangensis, 2025, 37(2): 338-348. |
| [13] | HU Rui, MA Liya, WAN Qun, WANG Ya, CAO Yaoyao, SHAO Sicheng, GE Jing, WU Xiangwei, YU Xiangyang. Effect of growth-promoting bacteria on the degradation of thiamethoxam in Brassica rapa subsp. chinensis [J]. Acta Agriculturae Zhejiangensis, 2025, 37(2): 394-404. |
| [14] | XIAO Yumiao, MA Qiaomei, ZHANG Sifa, HE Yong, ZHAO Zhenqing. Effects of fish protein hydrolysate on the growth and photosynthetic characteristics of tomato seedlings [J]. Acta Agriculturae Zhejiangensis, 2025, 37(12): 2504-2515. |
| [15] | YAN Peiyu, ZHANG Shengyin, CHEN Liang, LIU Bin. Effects of water-fertilizer coupling on growth, yield and quality of tomato cultivated in facility [J]. Acta Agriculturae Zhejiangensis, 2025, 37(12): 2516-2524. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||